I mentioned hand sanitizer being adulterated in an early snippet. This time the cheaters have switched and are using 1-propanol as the ingredient. I feel that we need a chemistry lesson to understand all of this.

Image from Pixabay
Correct hand sanitizer is made from ethanol; also known as ethyl alcohol. In chemistry ethyl means 2 carbons (C) and the “-ol” at the end of the name means that is has a hydroxyl group which is oxygen attached to hydrogen and represented by “-OH”. As carbon must be attached to four other elements, the gaps are filled with hydrogens (H). So ethanol is CH3CH2OH.
[I can’t seem to do subscript with substack so there is an image below with the formulas of simple alcohols written out.]
The first adulterant was wood alcohol or methanol, also known as methyl alcohol. Methyl is one carbon. So methanol is CH3OH.
Propanol in the current adulterated hand sanitizer is also known as propyl alcohol. Propyl is three carbons. Yes, you guessed it: propanol has the chemical formula CH3CH2CH2OH. There is another form of propanol that is (CH3)2CHOH. These are isomers of each other. The first one is propan-1-ol and is the compound in the hand sanitizers being recalled this time.
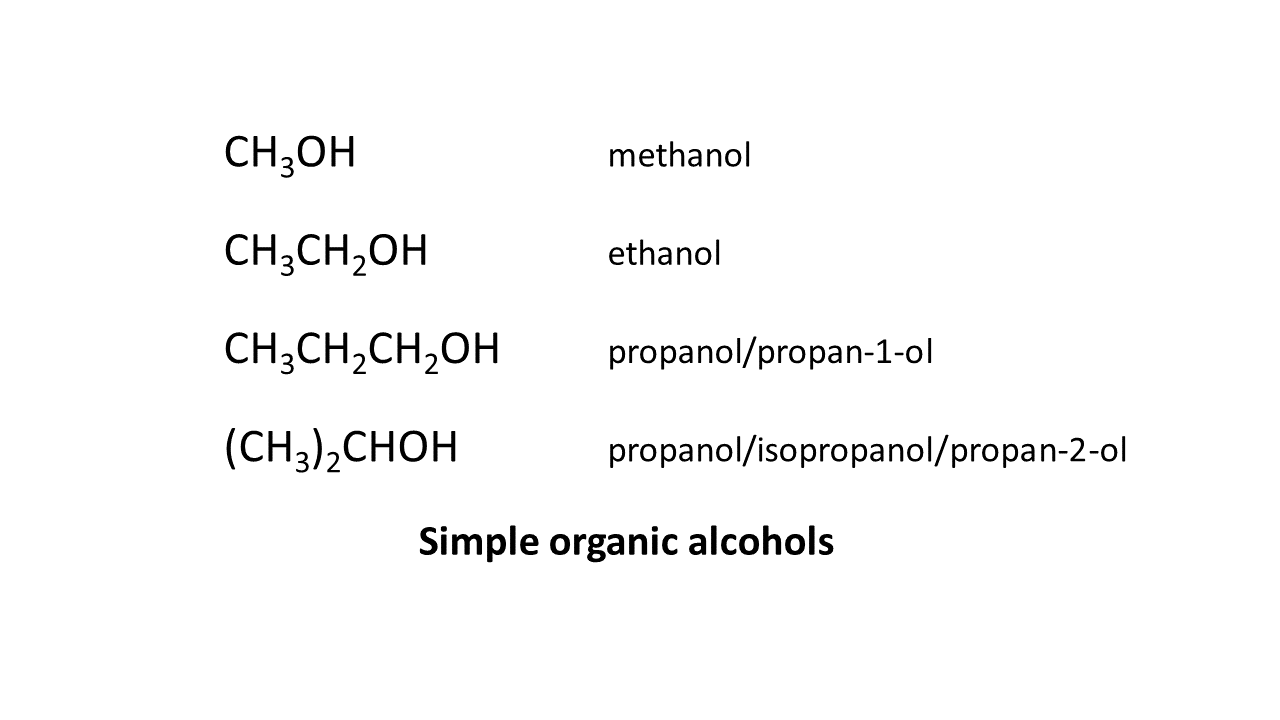
Image by Cathy Davies
That’s enough chemistry for today.
If you aren’t checking what hand sanitizers you are using, NOW might be a good time to start. Here is the list of hand sanitizers the FDA recommends you avoid. You need a receiving, storage and usage policy for chemical supplies as much as you do for receiving food ingredients. Schedule a free food safety chat to discuss what this might look like and make sure you are using chemicals such as hand sanitizers correctly and protecting your co-workers and customers.
Be safe. Wear a mask. Wash your damn hands. Eat a healthy diet.
Remember: Food Safety First. Non-compliance = Death




